Functional DNA Nanotechnology
The unique properties of nanoparticles that vary with shape, surface morphology and inter-particle distance, such as plasmonic and quantum confinement effects, make them promising choices for applications ranging from catalysis to biomedicine. To realize the full potential of nanomaterials, we are exploring novel ways to use DNA for the fine control of their morphology, precise spatial control of the position, orientation, and distances between multiple nanoparticles, and dynamic temporal control of nanomaterial assembly in response to multiple stimuli. The resulting functional DNA nanomaterials display many unique optical and catalytic properties with promising biomedical applications:
For a general overview of this area, refer to the above slideshow and the following review articles:
I. DNA-encoded Control of Nanomaterial Morphologies and Resulting Functional Properties
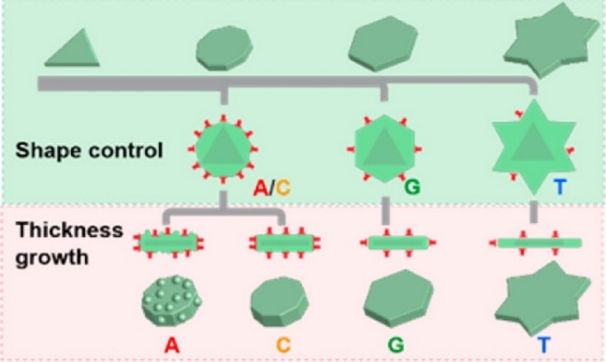
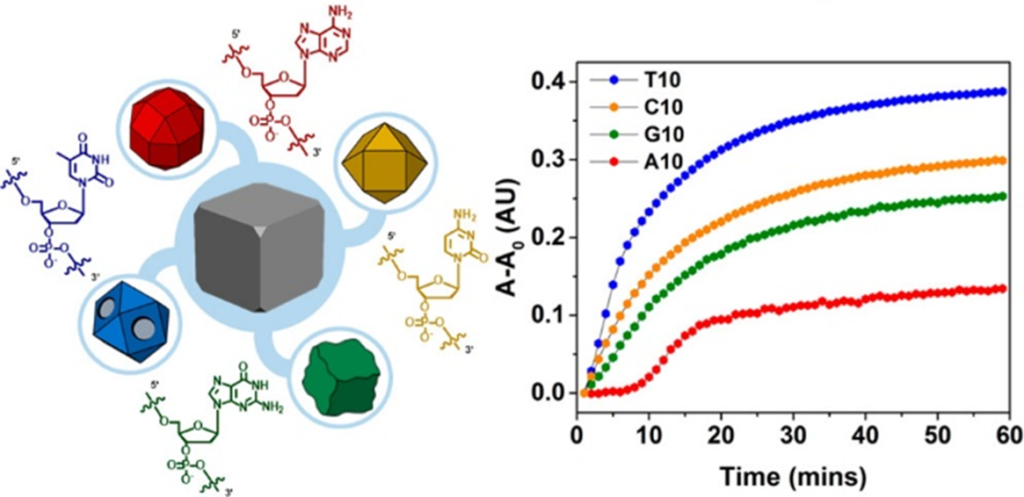
Just as nature uses genetic codes for precise control of protein synthesis, we are interested in discovering “genetic codes” (i.e., different combinations of DNA sequences) for controlling shapes and surface properties (i.e., morphologies) of nanomaterials. At the core of our endeavor is a systematic, predictive and customizable approach to synthesis of nanomaterials that brings the precision of living matter to materials design and engineering. Since the morphologies of nanoparticles are analogs of the three-dimensional structures of proteins from which the biological functions of the latter are derived, the programmable control of both monometallic and bimetallic nanoparticles has resulted in novel ways of fine-tuning optical and other properties of these nanomaterials while maintaining their colloidal stability.
II. Precise Spatial and Dynamic Temporal Control of Assembly of Nanomaterials and Biomolecules
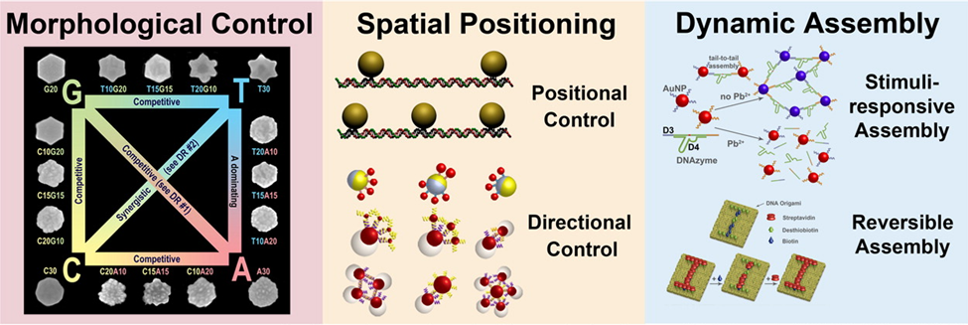
Nature has perfected the art of precise spatial and dynamic temporal control of biomaterials and biomolecules. Inspired by Nature, we are exploring methods to use DNA to control the assembly of materials or biomolecules. For precise spatial control, we have developed a method of using phosphorothioate DNA and bifunctional linkers to position nanoparticles and proteins into DNA origami templates with sub-nanometer resolution. For dynamic temporal control, we have used functional DNA (e.g., DNAzymes and aptamers) to control assembly and disassembly of gold nanoparticles, quantum dots, Janus nanoparticles, proteins and lanthanide-doped upconversion nanoparticles (UCNPs) in response to internal chemical and biological stimuli with cooperativity. Methods for proof reading and error corrections in nanomaterial assembly, as well as reversible assembly, have also been demonstrated.
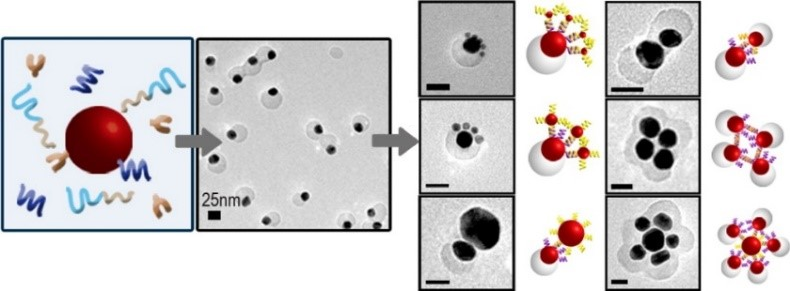
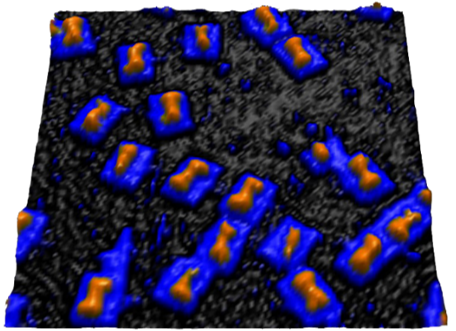
J. Am. Chem. Soc. 137, (16) 5272-5275 (2015), J. Am. Chem. Soc. 135, 2931-2934 (2013), J. Am. Chem. Soc. (135) 17675-17678 (2013), Chem. Sci. 4, (1) 398-404 (2012), ACS Nano 6, 802-809 (2012), Small 7, 1427-1430 (2011), J. Am. Chem. Soc. 132, 8906-8908 (2010)
III. Functional DNA-Based Nanomaterials for Biomedical Applications
While nanomaterials display many unique optical and catalytic properties, they often lack selectivity in these applications. By integrating functional DNA (e.g., DNAzymes and aptamers) with nanomaterials, including gold nanoparticles, quantum dots, iron oxide nanoparticles, polymer nanoparticles, upconversion nanoparticles (UCNPs), and liposomes, we have developed a novel class of colorimetric, fluorescent, and MRI sensors, as well as imaging and theranostic agents, aimed at detection and treatment of toxic chemicals and diseases, such as cancer.
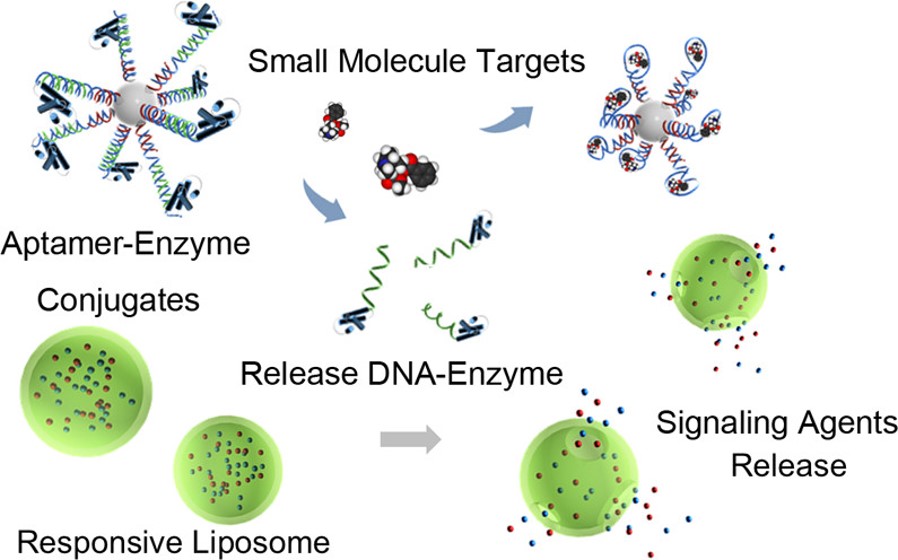
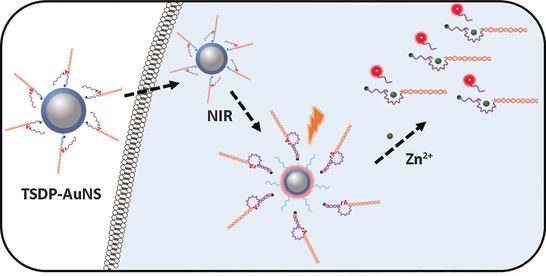
IV. Other Nanomaterials Projects
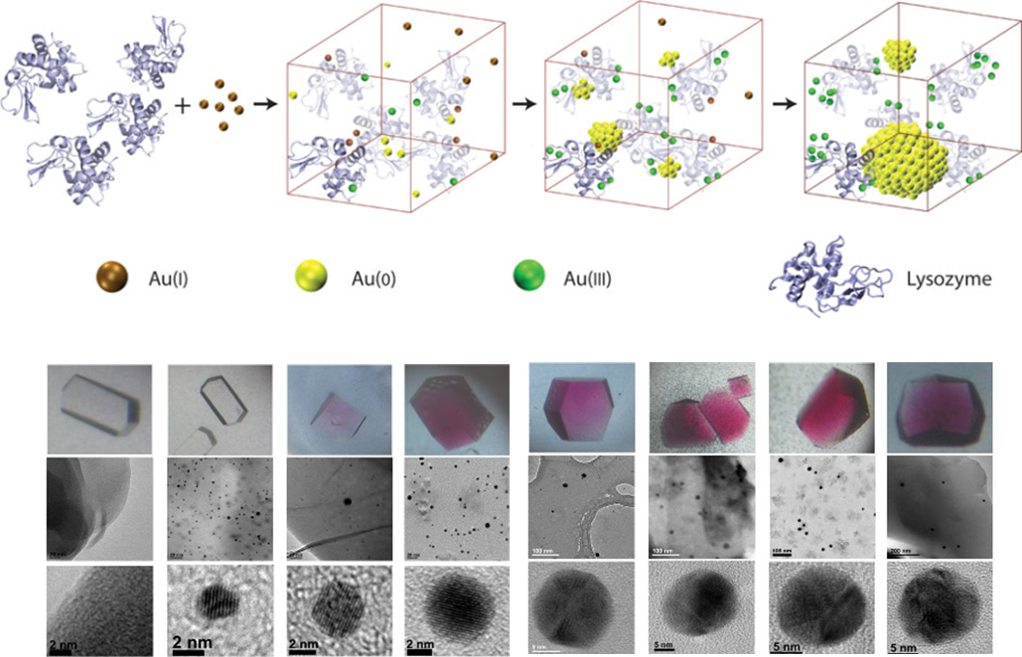
We are also interested in elucidating the mechanisms of nanoparticle or quantum dot formation in biomolecular templates such as protein crystals. Insights into elusive nanocrystal formation can be deduced from these studies. This project utilizes expertise from the interdisciplinary subgroups in the Lu lab and involves skills in both protein crystallization and nanoparticle synthesis and characterization.